 "In addition, the recently enacted CAA secures long-term separate payment for OMIDRIA in both ambulatory surgery centers and hospital outpatient departments, and we look forward to continued participation in OMIDRIA’s growth through our ongoing royalty stream." Demopulos, M.D., Omeros’ chairman and chief executive officer. The addition of the $200 million milestone payment from Rayner is expected to provide funding for Omeros’ operations across our pipeline well into at least 2025," said Gregory A. "Omeros ended 2022 with approximately $195 million available for operations. OMIDRIA sales are forecast to continue growing, in part as a result of the Consolidated Appropriations Act (CAA) of 2023, which expressly provides for separate payment of non-opioid pain management drugs, like OMIDRIA, in the outpatient surgery setting until January 1, 2028. net sales of OMIDRIA in 2022 totaled $131 million. sales of OMIDRIA on a country-by-country basis. Rayner is also obligated to pay a royalty of 15 percent on ex-U.S. net sales following achievement of the Milestone Event. net sales of OMIDRIA decreased from 50 percent to 30 percent of U.S. Per the terms of the Agreement, the royalty rate applicable to U.S. Rayner will continue to pay Omeros royalties on both U.S. Full payment owed by Rayner – the milestone payment and accrued interest – was received on February 3, 2023. The Milestone Event, as defined in the Agreement, occurred in late December 2022, entitling Omeros to receive the milestone payment of $200 million within 30 days thereafter. 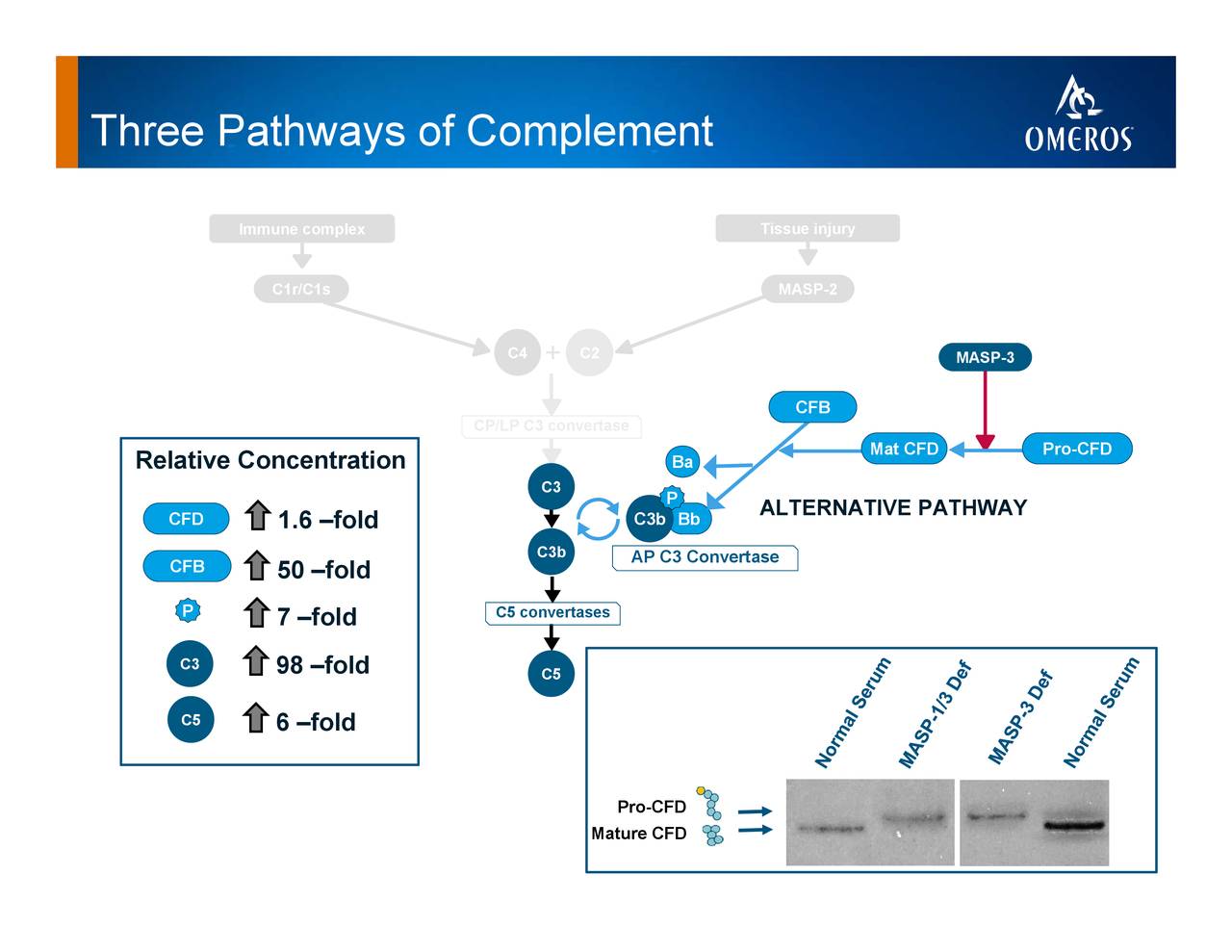
("Rayner") has paid the $200 million milestone payment due to Omeros under the Asset Purchase Agreement, dated Decem(the "Agreement"), pursuant to which Omeros sold its ophthalmology product OMIDRIA ® to Rayner in December of 2021. 
SEATTLE, February 06, 2023-( BUSINESS WIRE)-Omeros Corporation (Nasdaq: OMER) today announced that Rayner Surgical, Inc. 
One of these novel targets, GPR174, modulates a new cancer immunity axis recently discovered by Omeros, and the company is advancing GPR174-targeting antibodies and small-molecule inhibitors.– OMIDRIA ®-Related Payment Expected to Provide Operating Capital into at Least 2025 – Omeros' pipeline holds a diverse group of preclinical programs including a proprietary-asset-enabled antibody-generating technology and a proprietary GPCR platform through which it controls 54 GPCR drug targets and their corresponding compounds. OMS906, Omeros' inhibitor of MASP-3, the key activator of the alternative pathway of complement, is in a Phase 1 clinical trial, and the company's PDE7 inhibitor program OMS527, targeting addiction and movement disorders, has successfully completed a Phase 1 trial. Narsoplimab is also in multiple late-stage clinical development programs focused on other complement-mediated disorders, including IgA nephropathy, atypical hemolytic uremic syndrome and COVID-19. Omeros' lead MASP-2 inhibitor narsoplimab targets the lectin pathway of complement and is the subject of a biologics license application under priority review by FDA for the treatment of hematopoietic stem cell transplant-associated thrombotic microangiopathy. Its commercial product OMIDRIA® (phenylephrine and ketorolac intraocular solution) 1%/0.3% continues to gain market share in cataract surgery. Omeros is a commercial-stage biopharmaceutical company committed to discovering, developing and commercializing small-molecule and protein therapeutics for large-market and orphan indications targeting inflammation, complement-mediated diseases, disorders of the central nervous system and immune-related diseases, including cancers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
